.png)
Pacemakers Market By Product (external Pacemakers And Implantable Pacemakers) - Global Industry Analysis And Forecast To 2027
Published On : July 2018 Pages : 180 Category: Medical Devices Report Code : HC071131
SEGMENTS & REGIONS:
- By Product: external Pacemakers And Implantable Pacemakers
- Regions: North America, Europe, Asia- Pacific, Latin America, Middle East & Africa
Industry Trend Analysis
Increasing cases of Cardiovascular Diseases (CVDs), technological advancement in healthcare sectors and increasing reimbursement initiatives by the Government will primarily drive the growth of the pacemakers market. The increasing mortality rates due to inadequate treatment of cardiac diseases will encourage people to use artificial heart rate management devices owing to which the market is expected to have a steady growth in the upcoming years. For instance, as per CDC, in US, more than half of the Americans suffer from heart diseases every year including both men and women. This statistics indicate that artificial heart management device is the most commonly used device in this region. In addition, availability of adequate reimbursement facilities for implantation of pacemaker aids in reducing financial burden on patients thereby enhances the usage rates of these devices. Technological advancements in cardiac pacemaker are another positive aspect driving the market growth. Transitional tissue welding, dynamic peacemaking technology and microprocessor controlled devices are some of the advanced features that has been incorporated in the devices. However, high pricing of the devices often becomes unaffordable by the people despite of reimbursement facilities. The limitations of cardiac resynchronization therapy (CRT) pacemaker in pediatrics and risk of potential complications will impede the growth of the market. Thus, considering these drivers and restrains the pacemakers market is expected to exhibit steady growth over the forecast period.
Pacemakers Market Outlook and Trend Analysis
A pacemaker is a small, low-voltage medical device implanted in the chest or abdomen to help manage irregular heartbeats. It monitors how slow or fast heart beats and the pattern in which the heart beats. When the heart beats too slowly, the pacing device provides electrical stimulation. This device helps to reduce symptoms of fatigue and dizziness due to slow heart rhythm. During pacemaker implantation, a thin insulated wire, lead, is placed through the veins and into the heart. The lead’s tip is connected to the heart tissue and the other end is attached to the pacing device. The lead provides electrical pulses from the pacing device to the heart and transmits information from the heart back to the device. Around 4 million people across the globe have pacing device implanted. Some pacemakers are designed to facilitate patients to safely undergo an MRI scan. These are known as MRI ready or MR-conditional pacemakers.
Regional Outlook and Trend Analysis
North America held a significant share of the pacemaker market in 2017 due to the increasing incidence of cardiac diseases and technological advancement in healthcare sectors. The Asia-Pacific market has not experienced any substantial growth due to lack of large scale adoption of artificial heart management devices but can experience favorable growth in coming years owing to increasing awareness related to cardiac diseases in the region.
Competitive Outlook and Trend Analysis
The key players operating in the global pacemakers market emphasize on product development in order to introduce improved artificial cardiac management devices and capture a larger share of the market. Some of the major players in this market are Medtronic plc, St. Jude Medical, Biotronik, LivaNova PLC, Vitatron Holding B.V, Boston Scientific Corporation, MEDICO S.p.A and Pacetronix.
Market Opportunities
The key opportunity for the players operating in pacemakers market lies in the development of various types of cost-effective pacemakers and increasing awareness towards cardiac diseases. Apart from that, emergence of various advanced features in artificial cardiac management devices will further broaden the opportunities of the key players to grow significantly over the forecast period. In addition, promotional activities related to product information and benefits in daily lives of individuals, will further increase demand of this market, creating a lucrative opportunities for growth, for the key players of this market.
Pacemakers Market Segmentation:
By Product Type
- External Pacemakers
- Implantable Pacemakers
- Dual Chamber Pacemakers
- Single Chamber Pacemakers
- Biventricular Pacemakers
By Region
- North America
- U.S.
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Russia
- Italy
- Rest of Europe
- Asia-Pacific
- China
- Japan
- South Korea
- India
- Southeast Asia
- Rest of Asia-Pacific
- South America
- Brazil
- Argentina
- Columbia
- South Africa
- Rest of South America
- Middle East and Africa
- Saudi Arabia
- UAE
- Egypt
- Nigeria
- South Africa
- Rest of MEA
Some of the key questions answered by the report are:
- What was the market size in 2017 and forecast from 2017 to 2027?
- What will be the industry market growth from 2017 to 2027?
- What are the major drivers, restraints, opportunities, challenges, and industry trends and their impact on the market forecast?
- What are the major segments leading the market growth and why?
- Which are the leading players in the market and what are the major strategies adopted by them to sustain the market competition?
Market Classification
· Pacemakers Market, By Product, Estimates and Forecast, 2017-2027 ($Billion)
· External Pacemakers
· Implantable Pacemakers
o Dual Chamber Pacemakers
o Single Chamber Pacemakers
o Biventricular Pacemakers
· Pacemakers Market, By Region, Estimates and Forecast, 2017-2027 ($Billion)
o North America
§ North America Pacemakers Market, By Country
o U.S. Pacemakers Market
o Canada Pacemakers Market
o Mexico Pacemakers Market
o Europe
§ Europe Pacemakers Market, By Country
o Germany Pacemakers Market
o UK Pacemakers Market
o France Pacemakers Market
o Russia Pacemakers Market
o Italy Pacemakers Market
o Rest of Europe Pacemakers Market
o Asia-Pacific
§ Asia-Pacific Pacemakers Market, By Country
o China Pacemakers Market
o Japan Pacemakers Market
o South Korea Pacemakers Market
o India Pacemakers Market
o Southeast Asia Pacemakers Market
o Rest of Asia-Pacific Pacemakers Market
o South America
§ South America Pacemakers Market
o Brazil Pacemakers Market
o Argentina Pacemakers Market
o Columbia Pacemakers Market
o South Africa Pacemakers Market
o Rest of South America Pacemakers Market
o Middle East and Africa
§ Middle East and Africa Pacemakers Market
o Saudi Arabia Pacemakers Market
o UAE Pacemakers Market
o Egypt Pacemakers Market
o Nigeria Pacemakers Market
o South Africa Pacemakers Market
o Rest of MEA Pacemakers Market
Table of Contents
1. Introduction
2. Executive Summary
2.1. Key Highlights
3. Market Overview
3.1. Introduction
3.1.1. Market Definition
3.1.2. Market Segmentation
3.2. Market Dynamics
3.2.1. Drivers
3.2.1.1. Rising Prevalence of Cardiac vascular diseases (CVDs)
3.2.1.2. Increasing Reimbursement Initiatives by Government
3.2.1.3. Technological Advancement in Healthcare Sectors
3.2.2. Restraints
3.2.2.1. Lack of Skilled Professional for Handling The Device
3.2.2.2. High Maintenance Cost of the Device
3.2.2.3. Risk of Getting Potential Complications
3.2.3. Opportunities
3.2.3.1. Increased Application of Artificial Pancreas and Home Infusion Therapy
4. Market Analysis by Regions
4.1. North America (United States, Canada and Mexico)
4.1.1. United States Market States and Outlook (2017-2027)
4.1.2. Canada Market States and Outlook (2017-2027)
4.1.3. Mexico Market States and Outlook (2017-2027)
4.2. Europe (Germany, France, UK, Russia, Italy and Rest of Europe)
4.2.1. Germany Market States and Outlook (2013-2027)
4.2.2. France Market States and Outlook (2013-2027)
4.2.3. UK Market States and Outlook (2013-2027)
4.2.4. Russia Market States and Outlook (2013-2027)
4.2.5. Italy Market States and Outlook (2013-2027)
4.2.6. Rest of Europe Market States and Outlook (2013-2027)
4.3. Asia-Pacific(China, Japan, Korea, India, Southeast Asia and Rest of Asia-Pacific)
4.3.1. China Market States and Outlook (2013-2027)
4.3.2. Japan Market States and Outlook (2013-2027)
4.3.3. Korea Market States and Outlook (2013-2027)
4.3.4. India Market States and Outlook (2013-2027)
4.3.5. Southeast Asia Market States and Outlook (2013-2027)
4.3.6. Rest of Asia-Pacific Market States and Outlook (2013-2027)
4.4. South America (Brazil, Argentina, Columbia and Rest of South America)
4.4.1. Brazil Market States and Outlook (2013-2027)
4.4.2. Argentina Market States and Outlook (2013-2027)
4.4.3. Columbia Market States and Outlook (2013-2027)
4.4.4. Rest of South America Market States and Outlook (2013-2027)
4.5. Middle East and Africa (Saudi Arabia, UAE, Egypt, Nigeria, South Africa and Rest of MEA)
4.5.1. Saudi Arabia Market States and Outlook (2013-2027)
4.5.2. United Arab Emirates Market States and Outlook (2013-2027)
4.5.3. Egypt Market States and Outlook (2013-2027)
4.5.4. Nigeria Market States and Outlook (2013-2027)
4.5.5. South Africa Market States and Outlook (2013-2027)
4.5.6. Turkey Market States and Outlook (2013-2027)
4.5.7. Rest of Middle East and Africa Market States and Outlook (2013-2027)
5. Pacemakers Market, By Product
5.1. Introduction
5.2. Global Pacemakers Sales, Revenue and Market Share by Product (2013-2027)
5.2.1. Global Pacemakers Sales and Sales Share by Product (2013-2027)
5.2.2. Global Pacemakers Revenue and Revenue Share by Product (2013-2027)
5.3. External Pacemakers
5.3.1. Global External Pacemakers Sales and Growth Rate (2013-2027)
5.4. Implantable Pacemakers
5.4.1. Global Implantable Pacemakers Sales and Growth Rate (2013-2027)
5.4.2. Global Implantable Pacemakers By Type (2013-2027)
5.4.3. Dual Chamber Pacemakers
5.4.3.1. Global Dual Chamber Pacemakers Sales and Growth Rate (2013-2027)
5.4.4. Single Chamber Pacemakers
5.4.4.1. Global Single Chamber Pacemakers Sales and Growth Rate (2013-2027)
5.4.5. Biventricular Pacemakers
5.4.5.1. Global Biventricular Pacemakers Sales and Growth Rate (2013-2027)
6. Pacemakers Market, By Region
6.1. Introduction
6.2. Global Pacemakers Sales, Revenue and Market Share by Regions
6.2.1. Global Pacemakers Sales by Regions (2013-2027)
6.2.2. Global Pacemakers Revenue by Regions (2013-2027)
6.3. North America Pacemakers by Countries
6.3.1. North America Pacemakers Sales and Growth Rate (2013-2027)
6.3.2. North America Pacemakers Revenue and Growth Rate (2013-2027)
6.3.3. North America Pacemakers Sales by Countries (2013-2027)
6.3.4. North America Pacemakers Revenue (Million USD) by Countries (2013-2027)
6.3.5. United States
6.3.5.1. United States Pacemakers Sales and Growth Rate (2013-2027)
6.3.5.2. United States Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.3.6. Canada
6.3.6.1. Canada Pacemakers Sales and Growth Rate (2013-2027)
6.3.6.2. Canada Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.3.7. Mexico
6.3.7.1. Mexico Pacemakers Sales and Growth Rate (2013-2027)
6.3.7.2. Mexico Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.4. Europe Pacemakers by Countries
6.4.1. Europe Pacemakers Sales and Growth Rate (2013-2027)
6.4.2. Europe Pacemakers Revenue and Growth Rate (2013-2027)
6.4.3. Europe Pacemakers Sales by Countries (2013-2027)
6.4.4. Europe Pacemakers Revenue (Million USD) by Countries (2013-2027)
6.4.5. Germany
6.4.5.1. Germany Pacemakers Sales and Growth Rate (2013-2027)
6.4.5.2. Germany Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.4.6. France
6.4.6.1. France Pacemakers Sales and Growth Rate (2013-2027)
6.4.6.2. France Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.4.7. UK
6.4.7.1. UK Pacemakers Sales and Growth Rate (2013-2027)
6.4.7.2. UK Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.4.8. Russia
6.4.8.1. Russia Pacemakers Sales and Growth Rate (2013-2027)
6.4.8.2. Russia Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.4.9. Italy
6.4.9.1. Italy Pacemakers Sales and Growth Rate (2013-2027)
6.4.9.2. Italy Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.4.10. Rest of Europe
6.4.10.1. Rest of Europe Pacemakers Sales and Growth Rate (2013-2027)
6.4.10.2. Rest of Europe Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.5. Asia-Pacific Pacemakers by Countries
6.5.1. Asia-Pacific Pacemakers Sales and Growth Rate (2013-2027)
6.5.2. Asia-Pacific Pacemakers Revenue and Growth Rate (2013-2027)
6.5.3. Asia-Pacific Pacemakers Sales by Countries (2013-2027)
6.5.4. Asia-Pacific Pacemakers Revenue (Million USD) by Countries (2013-2027)
6.5.5. China
6.5.5.1. China Pacemakers Sales and Growth Rate (2013-2027)
6.5.5.2. China Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.5.6. Japan
6.5.6.1. Japan Pacemakers Sales and Growth Rate (2013-2027)
6.5.6.2. Japan Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.5.7. Korea
6.5.7.1. Korea Pacemakers Sales and Growth Rate (2013-2027)
6.5.7.2. Korea Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.5.8. India
6.5.8.1. India Pacemakers Sales and Growth Rate (2013-2027)
6.5.8.2. India Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.5.9. Southeast Asia
6.5.9.1. Southeast Asia Pacemakers Sales and Growth Rate (2013-2027)
6.5.9.2. Southeast Asia Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.5.10. Rest of Asia-Pacific
6.5.10.1. Rest of Asia-Pacific Pacemakers Sales and Growth Rate (2013-2027)
6.5.10.2. Rest of Asia-Pacific Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.6. South America Pacemakers by Countries
6.6.1. South America Pacemakers Sales and Growth Rate (2013-2027)
6.6.2. South America Pacemakers Revenue and Growth Rate (2013-2027)
6.6.3. South America Pacemakers Sales by Countries (2013-2027)
6.6.4. South America Pacemakers Revenue (Million USD) by Countries (2013-2027)
6.6.5. Brazil
6.6.5.1. Brazil Pacemakers Sales and Growth Rate (2013-2027)
6.6.5.2. Brazil Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.6.6. Argentina
6.6.6.1. Argentina Pacemakers Sales and Growth Rate (2013-2027)
6.6.6.2. Argentina Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.6.7. Columbia
6.6.7.1. Columbia Pacemakers Sales and Growth Rate (2013-2027)
6.6.7.2. Columbia Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.6.8. Rest of South America
6.6.8.1. Rest of South America Pacemakers Sales and Growth Rate (2013-2027)
6.6.8.2. Rest of South America Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.7. Middle East and Africa Pacemakers by Countries
6.7.1. Middle East and Africa Pacemakers Sales and Growth Rate (2013-2027)
6.7.2. Middle East and Africa Pacemakers Revenue and Growth Rate (2013-2027)
6.7.3. Middle East and Africa Pacemakers Sales by Countries (2013-2027)
6.7.4. Middle East and Africa Pacemakers Revenue (Million USD) by Countries (2013-2027)
6.7.5. Saudi Arabia
6.7.5.1. Saudi Arabia Pacemakers Sales and Growth Rate (2013-2027)
6.7.5.2. Saudi Arabia Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.7.6. United Arab Emirates
6.7.6.1. United Arab Emirates Pacemakers Sales and Growth Rate (2013-2027)
6.7.6.2. United Arab Emirates Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.7.7. Egypt
6.7.7.1. Egypt Pacemakers Sales and Growth Rate (2013-2027)
6.7.7.2. Egypt Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.7.8. Nigeria
6.7.8.1. Nigeria Pacemakers Sales and Growth Rate (2013-2027)
6.7.8.2. Nigeria Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.7.9. South Africa
6.7.9.1. South Africa Pacemakers Sales and Growth Rate (2013-2027)
6.7.9.2. South Africa Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.7.10. Turkey
6.7.10.1. Turkey Pacemakers Sales and Growth Rate (2013-2027)
6.7.10.2. Turkey Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
6.7.11. Rest of Middle East and Africa
6.7.11.1. Rest of Middle East and Africa Pacemakers Sales and Growth Rate (2013-2027)
6.7.11.2. Rest of Middle East and Africa Pacemakers Revenue (Millions USD) and Growth Rate (2013-2027)
7. Company Profiles
7.1. Medtronic plc.
7.1.1. Business Overview
7.1.2. Product Portfolio
7.1.3. Strategic Developments
7.1.4. Sales, Revenue and Market Share
7.2. St. Jude Medical
7.2.1. Business Overview
7.2.2. Product Portfolio
7.2.3. Strategic Developments
7.2.4. Sales, Revenue and Market Share
7.3. Biotronik
7.3.1. Business Overview
7.3.2. Product Portfolio
7.3.3. Strategic Developments
7.3.4. Sales, Revenue and Market Share
7.4. LivaNova PLC
7.4.1. Business Overview
7.4.2. Product Portfolio
7.4.3. Strategic Developments
7.4.4. Sales, Revenue and Market Share
7.5. Vitatron Holding B.V
7.5.1. Business Overview
7.5.2. Product Portfolio
7.5.3. Strategic Developments
7.5.4. Sales, Revenue and Market Share
7.6. Boston Scientific Corporation
7.6.1. Business Overview
7.6.2. Product Portfolio
7.6.3. Strategic Developments
7.6.4. Sales, Revenue and Market Share
7.7. MEDICO S.p.A
7.7.1. Business Overview
7.7.2. Product Portfolio
7.7.3. Strategic Developments
7.7.4. Sales, Revenue and Market Share
7.8. Pacetronix
7.8.1. Business Overview
7.8.2. Product Portfolio
7.8.3. Strategic Developments
7.8.4. Sales, Revenue and Market Share
8. Global Pacemakers Market Competition, by Manufacturer
8.1. Global Pacemakers Sales and Market Share by Manufacturer (2017-2017)
8.2. Global Pacemakers Revenue and Market Share by Manufacturer (2017-2017)
8.3. Global Pacemakers Price By Region (2017-2017)
8.4. Top 5 Pacemakers Manufacturer Market Share
8.5. Market Competition Trend
9. Pacemakers Market Forecast (2017-2027)
9.1. Global Pacemakers Sales, Revenue (Millions USD) and Growth Rate (2017-2027)
9.2. Pacemakers Market Forecast by Regions (2017-2027)
9.2.1. North America Pacemakers Market Forecast (2017-2027)
9.2.1.1. United States Pacemakers Market Forecast (2017-2027)
9.2.1.2. Canada Pacemakers Market Forecast (2017-2027)
9.2.1.3. Mexico Pacemakers Market Forecast (2017-2027)
9.2.2. Europe Pacemakers Market Forecast (2017-2027)
9.2.2.1. Germany Pacemakers Market Forecast (2017-2027)
9.2.2.2. France Pacemakers Market Forecast (2017-2027)
9.2.2.3. UK Pacemakers Market Forecast (2017-2027)
9.2.2.4. Russia Pacemakers Market Forecast (2017-2027)
9.2.2.5. Italy Pacemakers Market Forecast (2017-2027)
9.2.2.6. Rest of Europe Pacemakers Market Forecast (2017-2027)
9.2.3. Asia-Pacific Pacemakers Market Forecast (2017-2027)
9.2.3.1. China Pacemakers Market Forecast (2017-2027)
9.2.3.2. Japan Pacemakers Market Forecast (2017-2027)
9.2.3.3. Korea Pacemakers Market Forecast (2017-2027)
9.2.3.4. India Pacemakers Market Forecast (2017-2027)
9.2.3.5. Southeast Asia Pacemakers Market Forecast (2017-2027)
9.2.3.6. Rest of Asia-Pacific Pacemakers Market Forecast (2017-2027)
9.2.4. South America Pacemakers Market Forecast (2017-2027)
9.2.4.1. Brazil Pacemakers Market Forecast (2017-2027)
9.2.4.2. Argentina Pacemakers Market Forecast (2017-2027)
9.2.4.3. Columbia Pacemakers Market Forecast (2017-2027)
9.2.4.4. Rest of South America Pacemakers Market Forecast (2017-2027)
9.2.5. Middle East and Africa Pacemakers Market Forecast (2017-2027)
9.2.5.1. Saudi Arabia Pacemakers Market Forecast (2017-2027)
9.2.5.2. United Arab Emirates Pacemakers Market Forecast (2017-2027)
9.2.5.3. Egypt Pacemakers Market Forecast (2017-2027)
9.2.5.4. Nigeria Pacemakers Market Forecast (2017-2027)
9.2.5.5. South Africa Pacemakers Market Forecast (2017-2027)
9.2.5.6. Turkey Pacemakers Market Forecast (2017-2027)
9.2.5.7. Rest of Middle East and Africa Pacemakers Market Forecast (2017-2027)
9.3. Pacemakers Market Forecast by Product(2017-2027)
9.3.1. Pacemakers Sales Forecast by Product(2017-2027)
9.3.2. Pacemakers Sales Market Share Forecast by Product(2017-2027)
10. Research Methodology
10.1. Secondary Research
10.2. Primary Research
List of Tables
Figure United States Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Canada Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Mexico Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Germany Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure France Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure UK Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Russia Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Italy Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Rest of Europe Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure China Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Japan Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Korea Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure India Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Southeast Asia Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Rest of Asia-Pacific Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Brazil Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Argentina Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Columbia Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Rest of South America Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Saudi Arabia Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure United Arab Emirates Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Egypt Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Nigeria Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure South Africa Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Turkey Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Figure Rest of Middle East and Africa Pacemakers Revenue (Million USD) and Growth Rate (2017-2025)
Table Global Pacemakers Sales and Sales Share by Product (2017-2018)
Table Global Pacemakers Revenue and Revenue Share by Product (2017-2018)
Figure Global External Pacemakers Sales and Growth Rate (2017-2018)
Figure Global Implantable Pacemakers Sales and Growth Rate (2017-2018)
Figure Global Dual Chamber Pacemakers Sales and Growth Rate (2017-2018)
Figure Global Single Chamber Pacemakers Sales and Growth Rate (2017-2018)
Figure Global Biventricular Pacemakers Sales and Growth Rate (2017-2018)
Table Global Pacemakers Sales by Regions (2017-2018)
Table Global Pacemakers Revenue by Regions (2017-2018)
Figure North America Pacemakers Sales and Growth Rate (2017-2018)
Figure North America Pacemakers Revenue and Growth Rate (2017-2018)
Figure North America Pacemakers Sales by Countries (2017-2018)
Figure North America Pacemakers Revenue (Million USD) by Countries (2017-2018)
Figure United States Pacemakers Sales and Growth Rate (2017-2018)
Figure United States Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Canada Pacemakers Sales and Growth Rate (2017-2018)
Figure Canada Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Mexico Pacemakers Sales and Growth Rate (2017-2018)
Figure Mexico Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Europe Pacemakers Sales and Growth Rate (2017-2018)
Figure Europe Pacemakers Revenue and Growth Rate (2017-2018)
Figure Europe Pacemakers Sales by Countries (2017-2018)
Figure Europe Pacemakers Revenue (Million USD) by Countries (2017-2018)
Figure Germany Pacemakers Sales and Growth Rate (2017-2018)
Figure Germany Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure France Pacemakers Sales and Growth Rate (2017-2018)
Figure France Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure UK Pacemakers Sales and Growth Rate (2017-2018)
Figure UK Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Russia Pacemakers Sales and Growth Rate (2017-2018)
Figure Russia Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Italy Pacemakers Sales and Growth Rate (2017-2018)
Figure Italy Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Rest of Europe Pacemakers Sales and Growth Rate (2017-2018)
Figure Rest of Europe Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Asia-Pacific Pacemakers Sales and Growth Rate (2017-2018)
Figure Asia-Pacific Pacemakers Revenue and Growth Rate (2017-2018)
Figure Asia-Pacific Pacemakers Sales by Countries (2017-2018)
Figure Asia-Pacific Pacemakers Revenue (Million USD) by Countries (2017-2018)
Figure China Pacemakers Sales and Growth Rate (2017-2018)
Figure China Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Japan Pacemakers Sales and Growth Rate (2017-2018)
Figure Japan Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Korea Pacemakers Sales and Growth Rate (2017-2018)
Figure Korea Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure India Pacemakers Sales and Growth Rate (2017-2018)
Figure India Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Southeast Asia Pacemakers Sales and Growth Rate (2017-2018)
Figure Southeast Asia Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Rest of Asia-Pacific Pacemakers Sales and Growth Rate (2017-2018)
Figure Rest of Asia-Pacific Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure South America Pacemakers Sales and Growth Rate (2017-2018)
Figure South America Pacemakers Revenue and Growth Rate (2017-2018)
Figure South America Pacemakers Sales by Countries (2017-2018)
Figure South America Pacemakers Revenue (Million USD) by Countries (2017-2018)
Figure Brazil Pacemakers Sales and Growth Rate (2017-2018)
Figure Brazil Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Argentina Pacemakers Sales and Growth Rate (2017-2018)
Figure Argentina Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Columbia Pacemakers Sales and Growth Rate (2017-2018)
Figure Columbia Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Rest of South America Pacemakers Sales and Growth Rate (2017-2018)
Figure Rest of South America Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Middle East and Africa Pacemakers Sales and Growth Rate (2017-2018)
Figure Middle East and Africa Pacemakers Revenue and Growth Rate (2017-2018)
Figure Middle East and Africa Pacemakers Sales by Countries (2017-2018)
Figure Middle East and Africa Pacemakers Revenue (Million USD) by Countries (2017-2018)
Figure Saudi Arabia Pacemakers Sales and Growth Rate (2017-2018)
Figure Saudi Arabia Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure United Arab Emirates Pacemakers Sales and Growth Rate (2017-2018)
Figure United Arab Emirates Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Egypt Pacemakers Sales and Growth Rate (2017-2018)
Figure Egypt Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Nigeria Pacemakers Sales and Growth Rate (2017-2018)
Figure Nigeria Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure South Africa Pacemakers Sales and Growth Rate (2017-2018)
Figure South Africa Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Turkey Pacemakers Sales and Growth Rate (2017-2018)
Figure Turkey Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Figure Rest of Middle East and Africa Pacemakers Sales and Growth Rate (2017-2018)
Figure Rest of Middle East and Africa Pacemakers Revenue (Millions USD) and Growth Rate (2017-2018)
Table Medtronic plc. Pacemakers Sales, Price, Revenue, Gross Margin and Market Share (2017-2017)
Table St. Jude Medical Pacemakers Sales, Price, Revenue, Gross Margin and Market Share (2017-2017)
Table Biotronik Pacemakers Sales, Price, Revenue, Gross Margin and Market Share (2017-2017)
Table LivaNova PLC Pacemakers Sales, Price, Revenue, Gross Margin and Market Share (2017-2017)
Table Vitatron Holding B.V Pacemakers Sales, Price, Revenue, Gross Margin and Market Share (2017-2017)
Table Boston Scientific Corporation Pacemakers Sales, Price, Revenue, Gross Margin and Market Share (2017-2017)
Table MEDICO S.p.A Pacemakers Sales, Price, Revenue, Gross Margin and Market Share (2017-2017)
Table Pacetronix Pacemakers Sales, Price, Revenue, Gross Margin and Market Share (2017-2017)
Figure Global Pacemakers Sales and Market Share by Manufacturer
Figure Global Pacemakers Revenue and Market Share by Manufacturer
Table Global Pacemakers Price by Region (2017-2017)
Figure Top 5 Pacemakers Manufacturer (Revenue) Market Share
Figure Market Competition Trend
Figure Global Pacemakers Sales, Revenue (Millions USD) and Growth Rate (2018-2025)
Table Pacemakers Market Forecast by Regions (2018-2025)
Figure North America Pacemakers Market Forecast (2018-2025)
Figure United States Pacemakers Market Forecast (2018-2025)
Figure Canada Pacemakers Market Forecast (2018-2025)
Figure Mexico Pacemakers Market Forecast (2018-2025)
Figure Europe Pacemakers Market Forecast (2018-2025)
Figure Germany Pacemakers Market Forecast (2018-2025)
Figure France Pacemakers Market Forecast (2018-2025)
Figure UK Pacemakers Market Forecast (2018-2025)
Figure Russia Pacemakers Market Forecast (2018-2025)
Figure Italy Pacemakers Market Forecast (2018-2025)
Figure Rest of Europe Pacemakers Market Forecast (2018-2025)
Figure Asia-Pacific Pacemakers Market Forecast (2018-2025)
Figure China Pacemakers Market Forecast (2018-2025)
Figure Japan Pacemakers Market Forecast (2018-2025)
Figure Korea Pacemakers Market Forecast (2018-2025)
Figure India Pacemakers Market Forecast (2018-2025)
Figure Southeast Asia Pacemakers Market Forecast (2018-2025)
Figure Rest of Asia-Pacific Pacemakers Market Forecast (2018-2025)
Figure South America Pacemakers Market Forecast (2018-2025)
Figure Brazil Pacemakers Market Forecast (2018-2025)
Figure Argentina Pacemakers Market Forecast (2018-2025)
Figure Columbia Pacemakers Market Forecast (2018-2025)
Figure Rest of South America Pacemakers Market Forecast (2018-2025)
Figure Middle East and Africa Pacemakers Market Forecast (2018-2025)
Figure Saudi Arabia Pacemakers Market Forecast (2018-2025)
Figure United Arab Emirates Pacemakers Market Forecast (2018-2025)
Figure Egypt Pacemakers Market Forecast (2018-2025)
Figure Nigeria Pacemakers Market Forecast (2018-2025)
Figure South Africa Pacemakers Market Forecast (2018-2025)
Figure Turkey Pacemakers Market Forecast (2018-2025)
Figure Rest of Middle East and Africa Pacemakers Market Forecast (2018-2025)
Figure Global Pacemakers Sales Forecast by Product (2018-2025)
Figure Global Pacemakers Sales Market Share Forecast by Product (2018-2025)
Figure Global Pacemakers Sales Forecast by Product (2018-2025)
Research Methodology
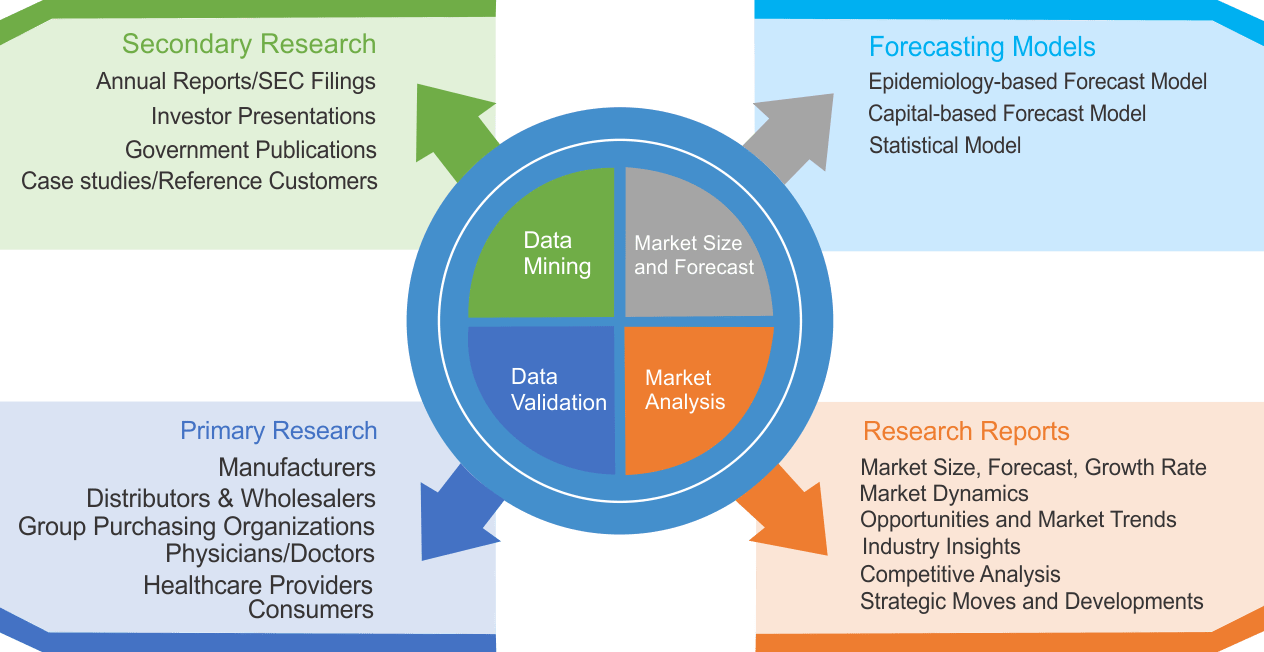
We use both primary as well as secondary research for our market surveys, estimates and for developing forecast. Our research process commence by analyzing the problem which enable us to design the scope for our research study. Our research process is uniquely designed with enough flexibility to adjust according to changing nature of products and markets, while retaining core element to ensure reliability and accuracy in research findings. We understand both macro and micro-economic factors to evaluate and forecast different market segments.
Data Mining
Data is extensively collected through various secondary sources such as annual reports, investor presentations, SEC filings, and other corporate publications. We also refer trade magazines, technical journals, paid databases such as Factiva and Bloomberg, industry trade journals, scientific journals, and social media data to understand market dynamics and industry trends. Further, we also conduct primary research to understand market drivers, restraints, opportunities, challenges, and competitive scenario to build our analysis.
Data Collection Matrix
|
Data Collection Matrix |
Supply Side |
Demand Side |
|
Primary Data Sources |
|
|
|
Secondary Data Sources |
|
|
Market Modeling and Forecasting
We use epidemiology and capital equipment-based models to forecast market size of different segments at country and regional level.
- Epidemiology-based Forecasting Model: This method uses epidemiology data gathered through various publications and from physicians to estimate population of patients, flow of treatment of individual disease and therapies. The data collected through this method includes statics on incidence of disease, population suffering from disease, and treatment population. This method is used to understand:
- Number of patients for particular device or medical procedure and
- Repeated use of particular device depending on health and condition of patient
- Capital-based Forecasting Model: This method of forecasting is based on number of replacements, installed-based and new sales of capital equipment used in various healthcare and diagnostic centers. These three parameters are calculated and forecast is developed. Installation base is calculated as average number of units per facility; while sales for particular year is calculated from number of new and replace units. Secondary data is collected through various supply chain intermediaries and opinion leaders to arrive at installation and sales rate. These techniques help our analysts in validating market and developed market estimates and forecast.
We do forecast on basis of several parameters such as market drivers, market opportunities, industry trends government regulations, raw materials supply and trade dynamics to ensure relevance of forecast with market scenario. With increasing need to granulized information, we used bottom-up methodology for forecasting where we evaluate each regional segment differently and combined all forecast to develop final market forecast.
Data Validation
We believe primary research is a very important tool in analyzing and forecasting different markets. In order to make sure accuracy of our findings, our team conducts primary interviews at every stage of research to gain deep insights into current business environment and future trends and key developments in market. This includes use of various methods such as telephonic interviews, focus groups, face to face interviews and questionnaires to validate our research from all aspects. We validate our data through primary research from key industry leaders such as CEO, product managers, marketing managers, suppliers, distributors, and consumers are frequently interviewed. These interviews provide valuable insights which help us to have better market understanding besides validating our estimates and forecast.
Data Triangulation
Industry Analysis
|
Qualitative Data |
Quantitative Data (2017-2025) |
|
|