Interventional Cardiology & Peripheral Vascular Devices Market By Product (angioplasty Stents, Angioplasty Balloon Catheter, Guidewires, Vascular Closure Devices, Peripheral Embolic Protection Devices, Atherectomy Devices, Endovascular Stent Grafts, Vascular Grafts, Thrombectomy Devices, Inferior Vena Cava Filter, Other Products) - Global Industry Analysis And Forecast To 2023
Published On : July 2018 Pages : 190 Category: In-Vitro Diagnostics Report Code : HC071100
Industry Outlook and Trend Analysis
The Interventional Cardiology & Peripheral Vascular Devices Market was worth USD 12.18 billion in 2017 and is expected to reach approximately USD 27.82 billion by 2023, while registering itself at a compound annual growth rate (CAGR) of 9.61% during the forecast period. Cardiovascular diseases is one of the essential driver of the worldwide mortality was in charge of 17.7 million passing’s around the world, as announced by the World Health Organization (WHO). This worldwide rate of death raised the significance of taking preventive wellbeing measures and cantering into advancement of enhanced treatment devices keeping in mind the end goal to accomplish powerful clinical results. Interventional method deployment in cardiology and fringe vascular ailment treatment is generally liked to stay away from entanglements and scar, diminishing torment and quicker recuperation.
Drivers & Restrains
Continuous mechanical developments in the field of interventional cardiology and fringe vascular gadgets are relied upon to drive the worldwide market. Researchers have concentrated on growing new frameworks with enhanced traits, for example, new age of medication eluting inflatables, and bio absorbable stents. Likewise, a few organizations have put resources into their research and development exercises to create imaginative products. Awareness among patients about coronary heart sicknesses and fringe vascular scatters assumes an imperative part in market development. A few instructive, government and non-legislative associations and key players have propelled awareness projects to expand the awareness level about the treatment of these sicknesses among patients, specialists, and human services suppliers. The effect of this factor as of now impacts the market development in the rising economies, as the appropriation of the propelled treatment device is low in the developing countries.
Regional Outlook and Trend Analysis
Over the previous decade, legislatures of different nations, particularly rising economies, have expanded their consumption on building up their human services framework. In this manner, increment in broad daylight and private use to build up the wellbeing framework in these locales will additionally support the selection of interventional cardiology and peripheral vascular devices in creating nations.
Competitive Insights
The leading players in the market are B. Braun Melsungen AG, Cardinal Health, Inc., Medtronic plc, Terumo Corporation, ABIOMED, Inc. and others. The major players in the market are profiled in detail in view of qualities, for example, company portfolio, business strategies, financial overview, recent developments, and share of the overall industry.
The Interventional Cardiology & Peripheral Vascular Devices Market is segmented as follows-
By Product:
- Angioplasty Stents
- Angioplasty Balloon Catheter
- Guidewires
- Vascular Closure Devices
- Peripheral Embolic Protection Devices
- Atherectomy Devices
- Endovascular Stent Grafts
- Vascular Grafts
- Thrombectomy Devices
- Inferior Vena Cava Filter
- Other Products
By Region
- North America
- U.S.
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Russia
- Italy
- Rest of Europe
- Asia-Pacific
- China
- Japan
- South Korea
- India
- Southeast Asia
- Rest of Asia-Pacific
- South America
- Brazil
- Argentina
- Columbia
- South Africa
- Rest of South America
- Middle East and Africa
- Saudi Arabia
- UAE
- Egypt
- Nigeria
- South Africa
- Rest of MEA
Some of the key questions answered by the report are:
- What was the market size in 2017 and forecast from 2017 to 2023?
- What will be the industry market growth from 2017 to 2023?
- What are the major drivers, restraints, opportunities, challenges, and industry trends and their impact on the market forecast?
- What are the major segments leading the market growth and why?
- Which are the leading players in the market and what are the major strategies adopted by them to sustain the market competition?
Market Classification
· Interventional Cardiology & Peripheral Vascular Devices Market, By Product, Estimates and Forecast, 2014-2023($Million)
· Angioplasty Stents
· Angioplasty Balloon Catheter
· Guidewires
· Vascular Closure Devices
· Peripheral Embolic Protection Devices
· Atherectomy Devices
· Endovascular Stent Grafts
· Vascular Grafts
· Thrombectomy Devices
· Inferior Vena Cava Filter
· Other Products
· Interventional Cardiology & Peripheral Vascular Devices Market, By Region, Estimates and Forecast, 2014-2023($Million)
o North America
§ North America Interventional Cardiology & Peripheral Vascular Devices Market, By Country
o U.S. Interventional Cardiology & Peripheral Vascular Devices Market
o Canada Interventional Cardiology & Peripheral Vascular Devices Market
o Mexico Interventional Cardiology & Peripheral Vascular Devices Market
o Europe
§ Europe Interventional Cardiology & Peripheral Vascular Devices Market, By Country
o Germany Interventional Cardiology & Peripheral Vascular Devices Market
o UK Interventional Cardiology & Peripheral Vascular Devices Market
o France Interventional Cardiology & Peripheral Vascular Devices Market
o Russia Interventional Cardiology & Peripheral Vascular Devices Market
o Italy Interventional Cardiology & Peripheral Vascular Devices Market
o Rest of Europe Interventional Cardiology & Peripheral Vascular Devices Market
o Asia-Pacific
§ Asia-Pacific Interventional Cardiology & Peripheral Vascular Devices Market, By Country
o China Interventional Cardiology & Peripheral Vascular Devices Market
o Japan Interventional Cardiology & Peripheral Vascular Devices Market
o South Korea Interventional Cardiology & Peripheral Vascular Devices Market
o India Interventional Cardiology & Peripheral Vascular Devices Market
o Southeast Asia Interventional Cardiology & Peripheral Vascular Devices Market
o Rest of Asia-Pacific Interventional Cardiology & Peripheral Vascular Devices Market
o South America
§ South America Interventional Cardiology & Peripheral Vascular Devices Market
o Brazil Interventional Cardiology & Peripheral Vascular Devices Market
o Argentina Interventional Cardiology & Peripheral Vascular Devices Market
o Columbia Interventional Cardiology & Peripheral Vascular Devices Market
o South Africa Interventional Cardiology & Peripheral Vascular Devices Market
o Rest of South America Interventional Cardiology & Peripheral Vascular Devices Market
o Middle East and Africa
§ Middle East and Africa Interventional Cardiology & Peripheral Vascular Devices Market
o Saudi Arabia Interventional Cardiology & Peripheral Vascular Devices Market
o UAE Interventional Cardiology & Peripheral Vascular Devices Market
o Egypt Interventional Cardiology & Peripheral Vascular Devices Market
o Nigeria Interventional Cardiology & Peripheral Vascular Devices Market
o South Africa Interventional Cardiology & Peripheral Vascular Devices Market
o Rest of MEA Interventional Cardiology & Peripheral Vascular Devices Market
Table of Contents
1. Introduction
1.1. Report Description
1.2. Research Methodology
1.2.1. Secondary Research
1.2.2. Primary Research
2. Executive Summary
2.1. Key Highlights
3. Market Overview
3.1. Introduction
3.1.1. Market Definition
3.1.2. Market Segmentation
3.2. Market Dynamics
3.2.1. Drivers
3.2.2. Restraints
3.2.3. Opportunities
3.2.3.1. Emerging Countries to Offer Lucrative Growth Opportunities
4. Market Analysis by Regions
4.1. North America (United States, Canada and Mexico)
4.1.1. United States Market States and Outlook (2017-2023)
4.1.2. Canada Market States and Outlook (2017-2023)
4.1.3. Mexico Market States and Outlook (2017-2023)
4.2. Europe (Germany, France, UK, Russia, Italy and Rest of Europe)
4.2.1. Germany Market States and Outlook (2017-2023)
4.2.2. France Market States and Outlook (2017-2023)
4.2.3. UK Market States and Outlook (2017-2023)
4.2.4. Russia Market States and Outlook (2017-2023)
4.2.5. Italy Market States and Outlook (2017-2023)
4.2.6. Rest of Europe Market States and Outlook (2017-2023)
4.3. Asia-Pacific (China, Japan, Korea, India, Southeast Asia and Rest of Asia-Pacific)
4.3.1. China Market States and Outlook (2017-2023)
4.3.2. Japan Market States and Outlook (2017-2023)
4.3.3. Korea Market States and Outlook (2017-2023)
4.3.4. India Market States and Outlook (2017-2023)
4.3.5. Rest of Asia-Pacific Market States and Outlook (2017-2023)
4.4. South America (Brazil, Argentina, Columbia and Rest of South America)
4.4.1. Brazil Market States and Outlook (2017-2023)
4.4.2. Argentina Market States and Outlook (2017-2023)
4.4.3. Columbia Market States and Outlook (2017-2023)
4.4.4. Rest of South America Market States and Outlook (2017-2023)
4.5. Middle East and Africa (Saudi Arabia, UAE, Egypt, Nigeria, South Africa and Rest of MEA)
4.5.1. Saudi Arabia Market States and Outlook (2017-2023)
4.5.2. UAE Market States and Outlook (2017-2023)
4.5.3. Egypt Market States and Outlook (2017-2023)
4.5.4. Nigeria Market States and Outlook (2017-2023)
4.5.5. South Africa Market States and Outlook (2017-2023)
4.5.6. Rest of MEA Market States and Outlook (2017-2023)
5. Interventional Cardiology & Peripheral Vascular Devices Market, By Product
5.1. Introduction
5.2. Global Interventional Cardiology & Peripheral Vascular Devices Sales, Revenue and Market Share by Product (2017-2027)
5.2.1. Global Interventional Cardiology & Peripheral Vascular Devices Sales and Sales Share by Product (2017-2027)
5.2.2. Global Interventional Cardiology & Peripheral Vascular Devices Revenue and Revenue Share by Product (2017-2027)
5.3. Angioplasty Stents
5.3.1. Global Angioplasty Stents Sales and Growth Rate (2017-2027)
5.4. Angioplasty Balloon Catheter
5.4.1. Global Angioplasty Balloon Catheter Sales and Growth Rate (2017-2027)
5.5. Guidewires
5.5.1. Global Guidewires Sales and Growth Rate (2017-2027)
5.6. Vascular Closure Devices
5.6.1. Global Vascular Closure Devices Sales and Growth Rate (2017-2027)
5.7. Peripheral Embolic Protection Devices
5.7.1. Global Peripheral Embolic Protection Devices Sales and Growth Rate (2017-2027)
5.8. Atherectomy Devices
5.8.1. Global Atherectomy Devices Sales and Growth Rate (2017-2027)
5.9. Endovascular Stent Grafts
5.9.1. Global Endovascular Stent Grafts Sales and Growth Rate (2017-2027)
5.10. Vascular Grafts
5.10.1. Global Vascular Grafts Sales and Growth Rate (2017-2027)
5.11. Thrombectomy Devices
5.11.1. Global Thrombectomy Devices Sales and Growth Rate (2017-2027)
5.12. Inferior Vena Cava Filter
5.12.1. Global Inferior Vena Cava Filter Sales and Growth Rate (2017-2027)
5.13. Other Products
5.13.1. Global Other Products Sales and Growth Rate (2017-2027)
6. Interventional Cardiology & Peripheral Vascular Devices Market, By Region
6.1. Introduction
6.2. Global Interventional Cardiology & Peripheral Vascular Devices Sales, Revenue and Market Share by Regions
6.2.1. Global Interventional Cardiology & Peripheral Vascular Devices Sales by Regions (2017-2027)
6.2.2. Global Interventional Cardiology & Peripheral Vascular Devices Revenue by Regions (2017-2027)
6.3. North America Interventional Cardiology & Peripheral Vascular Devices by Countries
6.3.1. North America Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.3.2. North America Interventional Cardiology & Peripheral Vascular Devices Revenue and Growth Rate (2017-2027)
6.3.3. North America Interventional Cardiology & Peripheral Vascular Devices Sales by Countries (2017-2027)
6.3.4. North America Interventional Cardiology & Peripheral Vascular Devices Revenue (Million USD) by Countries (2017-2027)
6.3.5. U.S.
6.3.5.1. United States Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.3.5.2. United States Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.3.6. Canada
6.3.6.1. Canada Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.3.6.2. Canada Interventional Cardiology & Peripheral Vascular Devices Sales (Millions USD) and Growth Rate (2017-2027)
6.3.7. Mexico
6.3.7.1. Mexico Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.3.7.2. Mexico Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.4. Europe Interventional Cardiology & Peripheral Vascular Devices by Countries
6.4.1. Europe Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.4.2. Europe Interventional Cardiology & Peripheral Vascular Devices Revenue and Growth Rate (2017-2027)
6.4.3. Europe Interventional Cardiology & Peripheral Vascular Devices Sales by Countries (2017-2027)
6.4.4. Europe Interventional Cardiology & Peripheral Vascular Devices Revenue (Million USD) by Countries (2017-2027)
6.4.5. Germany
6.4.5.1. Germany Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.4.5.2. Germany Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.4.6. UK
6.4.6.1. UK Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.4.6.2. UK Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.4.7. France
6.4.7.1. France Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.4.7.2. France Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.4.8. Russia
6.4.8.1. Russia Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.4.8.2. Russia Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.4.9. Italy
6.4.9.1. Italy Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.4.9.2. Italy Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.4.10. Rest of Europe
6.4.10.1. Rest of Europe Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.4.10.2. Rest of Europe Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.5. Asia-Pacific
6.5.1. Asia-Pacific Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.5.2. Asia-Pacific Interventional Cardiology & Peripheral Vascular Devices Revenue and Growth Rate (2017-2027)
6.5.3. Asia-Pacific Interventional Cardiology & Peripheral Vascular Devices Sales by Countries (2017-2027)
6.5.4. Asia-Pacific Interventional Cardiology & Peripheral Vascular Devices Revenue (Million USD) by Countries (2017-2027)
6.5.5. China
6.5.5.1. China Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.5.5.2. China Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.5.6. Japan
6.5.6.1. Japan Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.5.6.2. Japan Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.5.7. Korea
6.5.7.1. Korea Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.5.7.2. Korea Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.5.8. India
6.5.8.1. India Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.5.8.2. India Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.5.9. Southeast Asia
6.5.9.1. Southeast Asia Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.5.9.2. Southeast Asia Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.5.10. Rest of Asia-Pacific
6.5.10.1. Rest of Asia-Pacific Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.5.10.2. Rest of Asia-Pacific Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.6. South America
6.6.1. South America Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.6.2. South America Interventional Cardiology & Peripheral Vascular Devices Revenue and Growth Rate (2017-2027)
6.6.3. South America Interventional Cardiology & Peripheral Vascular Devices Sales by Countries (2017-2027)
6.6.4. South America Interventional Cardiology & Peripheral Vascular Devices Revenue (Million USD) by Countries (2017-2027)
6.6.5. Brazil
6.6.5.1. Brazil Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.6.5.2. Brazil Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.6.6. Argentina
6.6.6.1. Argentina Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.6.6.2. Argentina Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.6.7. Columbia
6.6.7.1. Columbia Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.6.7.2. Columbia Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.6.8. Rest of South America
6.6.8.1. Rest of South America Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.6.8.2. Rest of South America Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.7. Middle East and Africa
6.7.1. Middle East and Africa Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.7.2. Middle East and Africa Interventional Cardiology & Peripheral Vascular Devices Revenue and Growth Rate (2017-2027)
6.7.3. Middle East and Africa Interventional Cardiology & Peripheral Vascular Devices Sales by Countries (2017-2027)
6.7.4. Middle East and Africa Interventional Cardiology & Peripheral Vascular Devices Revenue (Million USD) by Countries (2017-2027)
6.7.5. Saudi Arabia
6.7.5.1. Saudi Arabia Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.7.5.2. Saudi Arabia Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.7.6. United Arab Emirates
6.7.6.1. United Arab Emirates Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.7.6.2. United Arab Emirates Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.7.7. Egypt
6.7.7.1. Egypt Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.7.7.2. Egypt Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.7.8. Nigeria
6.7.8.1. Nigeria Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.7.8.2. Nigeria Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.7.9. South Africa
6.7.9.1. South Africa Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.7.9.2. South Africa Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
6.7.10. Rest of Middle East and Africa
6.7.10.1. Rest of Middle East and Africa Interventional Cardiology & Peripheral Vascular Devices Sales and Growth Rate (2017-2027)
6.7.10.2. Rest of Middle East and Africa Interventional Cardiology & Peripheral Vascular Devices Revenue (Millions USD) and Growth Rate (2017-2027)
7. Company Profiles
7.1. Abbott Laboratories
7.1.1. Business Overview
7.1.2. Product Portfolio
7.1.3. Strategic Developments
7.1.4. Sales, Revenue and Market Share
7.2. B. Braun Melsungen AG
7.2.1. Business Overview
7.2.2. Product Portfolio
7.2.3. Strategic Developments
7.2.4. Sales, Revenue and Market Share
7.3. Baxter International Inc.
7.3.1. Business Overview
7.3.2. Product Portfolio
7.3.3. Strategic Developments
7.3.4. Sales, Revenue and Market Share
7.4. Boston Scientific Corporation
7.4.1. Business Overview
7.4.2. Product Portfolio
7.4.3. Strategic Developments
7.4.4. Sales, Revenue and Market Share
7.5. C. R. Bard, Inc.
7.5.1. Business Overview
7.5.2. Product Portfolio
7.5.3. Strategic Developments
7.5.4. Sales, Revenue and Market Share
7.6. Cardinal Health
7.6.1. Business Overview
7.6.2. Product Portfolio
7.6.3. Strategic Developments
7.6.4. Sales, Revenue and Market Share
7.7. Cook Medical, Inc.
7.7.1. Business Overview
7.7.2. Product Portfolio
7.7.3. Strategic Developments
7.7.4. Sales, Revenue and Market Share
7.8. Edwards Lifesciences Corporation
7.8.1. Business Overview
7.8.2. Product Portfolio
7.8.3. Strategic Developments
7.8.4. Sales, Revenue and Market Share
7.9. Medtronic plc.
7.9.1. Business Overview
7.9.2. Product Portfolio
7.9.3. Strategic Developments
7.9.4. Sales, Revenue and Market Share
7.10. St. Jude Medical
7.10.1. Business Overview
7.10.2. Product Portfolio
7.10.3. Strategic Developments
7.10.4. Sales, Revenue and Market Share
8. Global Interventional Cardiology & Peripheral Vascular Devices Market Competition, by Manufacturer
8.1. Global Interventional Cardiology & Peripheral Vascular Devices Sales and Market Share by Manufacturer (2017-2017)
8.2. Global Interventional Cardiology & Peripheral Vascular Devices Revenue and Market Share by Manufacturer (2017-2017)
8.3. Global Interventional Cardiology & Peripheral Vascular Devices Price by Manufacturer (2017-2017)
8.4. Top 5 Interventional Cardiology & Peripheral Vascular Devices Manufacturer Market Share
8.5. Market Competition Trend
9. Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.1. Global Interventional Cardiology & Peripheral Vascular Devices Sales, Revenue (Millions USD) and Growth Rate (2027-2023)
9.2. Interventional Cardiology & Peripheral Vascular Devices Market Forecast by Regions (2027-2023)
9.2.1. North America Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.1.1. United States Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.1.2. Canada Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.1.3. Mexico Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.2. Europe Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.2.1. Germany Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.2.2. United Kingdom Interventional Cardiology & Peripheral Vascular Devicess Market Forecast (2027-2023)
9.2.2.3. France Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.2.4. Russia Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.2.5. Italy Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.2.6. Rest of the Europe Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.3. Asia-Pacific Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.3.1. China Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.3.2. Japan Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.3.3. Korea Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.3.4. India Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.3.5. Southeast Asia Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.3.6. Rest of Asia-Pacific Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.4. South America Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.4.1. Brazil Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.4.2. Argentina Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.4.3. Columbia Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.4.4. Rest of South America Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.5. Middle East and Africa Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.5.1. Saudi Arabia Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.5.2. UAE Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.5.3. Egypt Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.5.4. Nigeria Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.5.5. South Africa Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.2.5.6. Rest of MEA Interventional Cardiology & Peripheral Vascular Devices Market Forecast (2027-2023)
9.3. Interventional Cardiology & Peripheral Vascular Devices Market Forecast by Product (2027-2023)
9.3.1. Global Interventional Cardiology & Peripheral Vascular Devices Sales Forecast by Product (2027-2023)
9.3.2. Global Interventional Cardiology & Peripheral Vascular Devices Sales Market Share Forecast by Product (2027-2023)
List of Tables
*You can glance through the list of Tables and Figures when you view the sample copy of Interventional Cardiology & Peripheral Vascular Devices Market.
Research Methodology
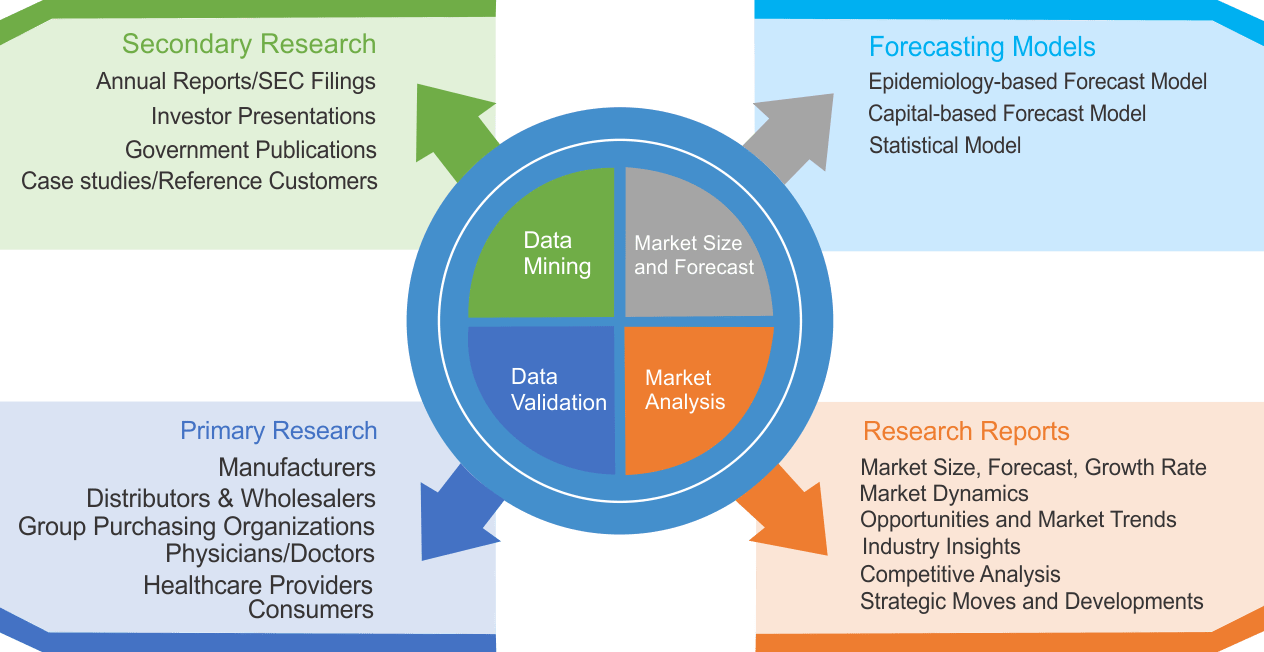
We use both primary as well as secondary research for our market surveys, estimates and for developing forecast. Our research process commence by analyzing the problem which enable us to design the scope for our research study. Our research process is uniquely designed with enough flexibility to adjust according to changing nature of products and markets, while retaining core element to ensure reliability and accuracy in research findings. We understand both macro and micro-economic factors to evaluate and forecast different market segments.
Data Mining
Data is extensively collected through various secondary sources such as annual reports, investor presentations, SEC filings, and other corporate publications. We also refer trade magazines, technical journals, paid databases such as Factiva and Bloomberg, industry trade journals, scientific journals, and social media data to understand market dynamics and industry trends. Further, we also conduct primary research to understand market drivers, restraints, opportunities, challenges, and competitive scenario to build our analysis.
Data Collection Matrix
|
Data Collection Matrix |
Supply Side |
Demand Side |
|
Primary Data Sources |
|
|
|
Secondary Data Sources |
|
|
Market Modeling and Forecasting
We use epidemiology and capital equipment-based models to forecast market size of different segments at country and regional level.
- Epidemiology-based Forecasting Model: This method uses epidemiology data gathered through various publications and from physicians to estimate population of patients, flow of treatment of individual disease and therapies. The data collected through this method includes statics on incidence of disease, population suffering from disease, and treatment population. This method is used to understand:
- Number of patients for particular device or medical procedure and
- Repeated use of particular device depending on health and condition of patient
- Capital-based Forecasting Model: This method of forecasting is based on number of replacements, installed-based and new sales of capital equipment used in various healthcare and diagnostic centers. These three parameters are calculated and forecast is developed. Installation base is calculated as average number of units per facility; while sales for particular year is calculated from number of new and replace units. Secondary data is collected through various supply chain intermediaries and opinion leaders to arrive at installation and sales rate. These techniques help our analysts in validating market and developed market estimates and forecast.
We do forecast on basis of several parameters such as market drivers, market opportunities, industry trends government regulations, raw materials supply and trade dynamics to ensure relevance of forecast with market scenario. With increasing need to granulized information, we used bottom-up methodology for forecasting where we evaluate each regional segment differently and combined all forecast to develop final market forecast.
Data Validation
We believe primary research is a very important tool in analyzing and forecasting different markets. In order to make sure accuracy of our findings, our team conducts primary interviews at every stage of research to gain deep insights into current business environment and future trends and key developments in market. This includes use of various methods such as telephonic interviews, focus groups, face to face interviews and questionnaires to validate our research from all aspects. We validate our data through primary research from key industry leaders such as CEO, product managers, marketing managers, suppliers, distributors, and consumers are frequently interviewed. These interviews provide valuable insights which help us to have better market understanding besides validating our estimates and forecast.
Data Triangulation
Industry Analysis
|
Qualitative Data |
Quantitative Data (2017-2025) |
|
|